  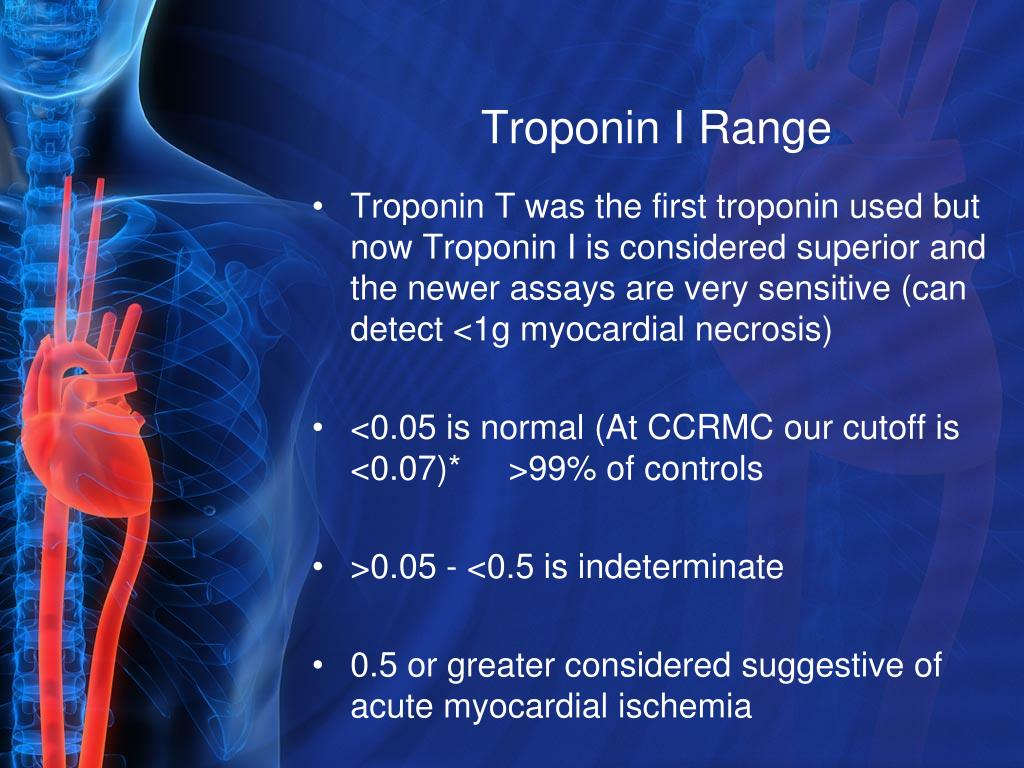
By acting as a circulating decoy for interleukin-33 (IL-33), it prevents the cardioprotective effects of the ST2/IL-33 signaling, thus promoting maladaptive myocardial hypertrophy and cardiomyocyte apoptosis. sST2 is mainly produced in extracardiac tissues in response to proinflammatory and profibrotic stimuli. Among them, soluble suppression of tumorigenesis-2 (sST2), a member of the interleukin 1 receptor family, emerged as an efficient prognostic biomarker for patients with chronic HF. Though the cardiac-specific biomarkers brain natriuretic peptide (BNP) and cardiac troponins (cTn) are firmly established as cornerstones of the diagnosis of heart failure (HF) and myocardial injury, respectively, a growing body of evidence supported the role of several non-cardiac specific biomarkers in describing the most relevant pathophysiological features of HF, i.e., inflammation, oxidative stress, extracellular matrix remodeling, neurohormonal activation, myocyte injury and stress. While their assessment has been shown to invariably improve the discrimination and reclassification of the risk of developing endpoints such as major adverse CV events (MACE), there is still uncertainty on whether their serial measurement may be useful to predict CV mortality or the cardioprotective benefits of T2DM medications. More recently, a role for cardiac biomarkers has been advocated in the setting of CV outcome trials in T2DM. Ī large body of evidence was published on the associations of single biomarkers with CV risk in T2DM, but simultaneous evaluations of large numbers of biomarkers in diabetic populations have been limited. Risk models for use in individuals with diabetes typically do not include information from biomarkers other than cholesterol, glycated hemoglobin (HbA1c), and urinary albumin-to-creatinine ratio (UACR), raising the question of whether adding novel biomarkers would improve CV risk prediction in patients with T2DM. The long-term prognostic value of sST2 and its ability to track variables related to insulin resistance and associated metabolic disorders support its implementation into routine clinical practice.Ĭurrent epidemiologic data have demonstrated that type 2 diabetes (T2DM) is accompanied by a 2–4-fold greater overall risk of cardiovascular (CV) complications compared to non-diabetic patients, even after adjustment for traditional risk factors. SST2, hs-cTnI, and NT-proBNP are associated with 15-year mortality and onset of CV events in T2DM. A ‘cardiac score’ based on the combination of sST2, hs-cTnI, and NT-proBNP was significantly associated with all-cause mortality (HR 1.35, C-statistic = 0.739) and development of CV events. 
SST2 followed an increasing trend from CTR to uncomplicated T2DM patients (T2DM-NC) to patients with at least one complication (T2DM-C), while hs-cTnI was significantly higher in T2DM-C compared to CTR but not to T2DM-NC. Their association with all-cause mortality and the development of diabetic complications was tested in T2DM patients over a median follow-up of 16.8 years using Cox models and logistic regressions. Serum levels of soluble suppression of tumorigenesis 2 (sST2), high-sensitivity (hs)-cTnI, and N-terminal (NT)-proBNP were assessed in 568 patients with T2DM and 115 healthy controls (CTR). Beyond the established role of brain natriuretic peptide (BNP) and cardiac troponins (cTn), other non-cardiac-specific biomarkers are emerging as predictors of CV outcomes in T2DM. Patients with type 2 diabetes (T2DM) present an increased risk of cardiovascular (CV) disease and excess CV-related mortality. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
